
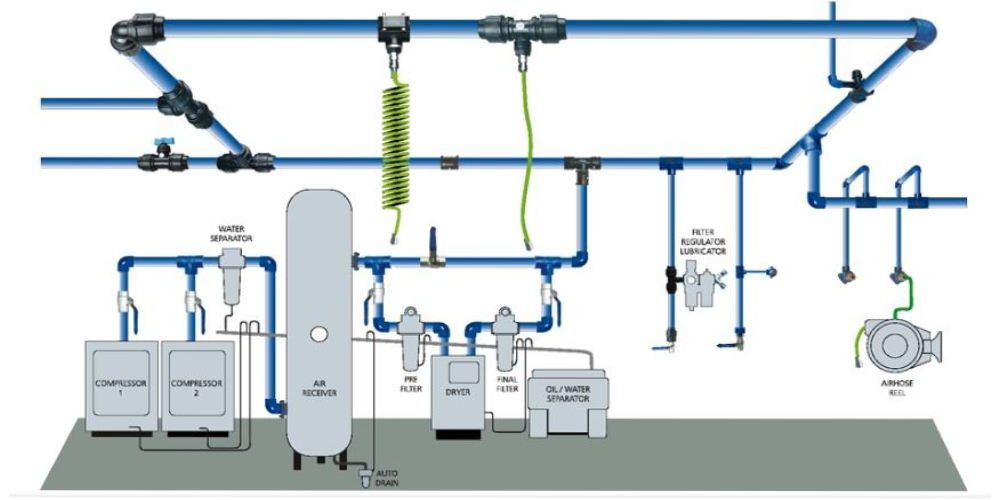
Compressed Air Validation is a critical quality assurance service used to verify that compressed air systems meet required purity, safety, and regulatory standards in GMP-controlled environments. In pharmaceutical, biotechnology, medical device, food, and cleanroom facilities, compressed air often comes in direct or indirect contact with products, equipment, and critical processes.
Compressed air validation ensures that the air used is free from contaminants such as particulates, oil vapors, moisture, and microorganisms that could compromise product quality or process integrity. The validation process involves systematic testing, monitoring, and documentation of compressed air quality in accordance with international GMP and ISO guidelines.
By performing compressed air validation, organizations can confirm that their compressed air systems consistently deliver air of the required quality, ensuring compliance with regulatory expectations and maintaining a controlled manufacturing environment.
1. Ensuring Product Quality
Compressed air is widely used in manufacturing processes such as material handling, equipment operation, cleaning, and packaging. If the compressed air contains contaminants, it can directly affect product purity and performance. Compressed Air Validation helps identify and control potential risks, ensuring that the final product meets quality, safety, and performance standards.
2. Protecting Patient Safety
In regulated industries like pharmaceuticals and healthcare, even minor contamination can pose serious risks to patient health. Validated compressed air systems help prevent the introduction of harmful particles, oil residues, or microbial contamination into products, thereby safeguarding patient safety and public health.
3. Regulatory Compliance
Regulatory authorities such as GMP, WHO, FDA, and EU GMP require manufacturers to validate all critical utilities, including compressed air. Compressed Air Validation demonstrates compliance with ISO 8573 standards and regulatory expectations, helping organizations pass audits and inspections with confidence.
4. Reducing Operational Risks
Unvalidated compressed air systems can lead to equipment damage, production downtime, and costly product recalls. Regular validation ensures system reliability, early detection of issues, and long-term operational efficiency.
5. Maintaining Cleanroom Integrity
In cleanroom environments, compressed air quality directly impacts cleanliness levels. Proper validation supports contamination control strategies and maintains the integrity of critical manufacturing areas.
| S.No | Test Parameters | Units |
|---|---|---|
| 1 | Dew Point | °C |
| 2 | Oil Mist | mg/m³ |
| 3 | Water Content | ppm v/v |
| 4 | Hydrocarbons | ppm v/v |
| 5 | Carbon Dioxide Conc. | ppm v/v |
| 6 | Carbon Monoxide Conc. | ppm v/v |
| 7 | Oxygen Concentration | % |
| 8 | Suspended Particulate Matter | mg/m³ |
| 9 | NO + NO2 | ppm v/v |
| 10 | Sulphur Dioxide | ppm v/v |
| 11 | Non Viable Particle Counter | µm |
| 12 | Total Microbial Count | Cfu/m³ |
H.No 47-1/C, 1St Floor, Sri Sainagar Colony, HMT Main Road, Chintal, Hyderabad - 500037 Telangana, INDIA
